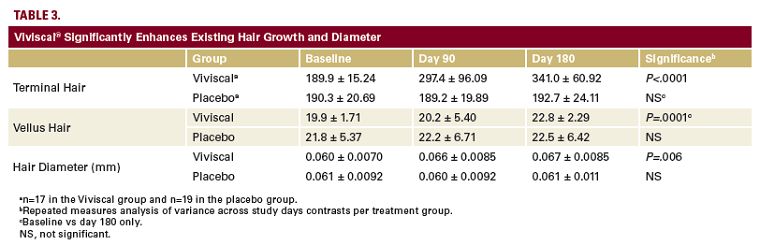
(range 24-55) and self-perceived thinning hair were enrolled and randomized in 2 groups.31 Changes in hair growth were measured using phototrichogram imaging of a 0.25 cm2 area of scalp. Hair type was defined by hair shaft diameter as vellus-like (≤40 μm) or terminal (>40 μm). Furthermore, hair shedding was assessed using a validated protocol that collected shed hairs during in-clinic shampooing.
After 6 months, Viviscal-treated subjects demonstrated a significant
increase in mean vellus-like hair caliber (diameter; Figure 2). This observation suggests that vellus hairs were transitioning
towards terminal hair classification, a process that may
continue to develop over the course of the treatment. Moreover,
subjects displayed a significant reduction (18.3%) in hair shedding after 3 months of daily treatment.

In yet another recent multi-site, double-blind, placebo-controlled
study, 40 women, 20 to 65 years of age, were randomized to receive either daily doses of Viviscal or a placebo for 6 months.32 Subjects’ scalps were photographed at baseline and after 6 months, and evaluated by an independent third party (Figure 3). An increase of 57% in hair count was observed in the treatment group after 3 months. Remarkably, the hair count further increased by 80% after 6 months of daily intake of Vivis






