bind alpha-gal epitopes and lead to rapid activation of complement
and cell lysis. The pre-existing anti-alpha-gal antibodies
found in most individuals are thought to be due to exposure to
alpha-gal epitopes that are naturally expressed on normal gut
flora, leading to chronic immunological stimulation.
In addition to the immune checkpoint therapy, half of the patients
will also receive dorgenmeltucel-L. Endpoints of the study include
safety assessments, efficacy, and immunological responses.

MELANOMA
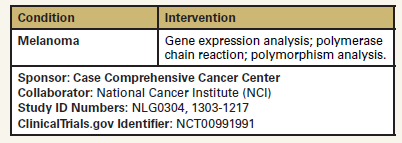
Study of Tumor Tissue Samples From Patients With Stage I, Stage II, or Stage III Malignant Melanoma
The objective of this study is to determine the genetic profile
of primary melanomas with and without synchronous regional
nodal involvement by examining for (1) activating mutations BRaf
and N-Ras associated with melanoma development, and (2)
allelic imbalances across the genome. This study will compare
the genetic profile of primary melanomas from patients with
and without lymph node involvement. It will also determine the
combinations of genetic lesions that correlate with nodal metastasis
by adopting a statistical machine learning approach to
build a lesion-based classifier for nodal metastasis.
Tumor tissue samples are collected from patients with stage I,
stage II, or stage III malignant melanoma. Laser capture microdissection
is performed on the archived tissue samples to isolate
melanoma cells. DNA is then purified from the samples and amplified
using polymerase chain reaction (PCR). Matrix-assisted
laser desorption/ionization (MALDI)-time of flight mass spectrometry
technology is used to detect mutations of B-Raf and N-Ras.
Single nucleotide polymorphism arrays are also performed.
VITILIGO
Assessing the Efficacy of Needling With or Without Corticosteroids in the Repigmentation of Vitiligo
This study is the first randomized control trial (RCT) of needling
in vitiligo that uses an objective measure to quantify results.
It has the potential to establish needling as a novel, effective
treatment for vitiligo, and to evaluate the use of confocal microscopy
(CFM) for monitoring response to treatment.
Vitiligo is an autoimmune cutaneous disorder that destroys melanocytes
leading to depigmented areas of skin. In the United
States, vitiligo affects 1% of patients, causing not only changes
in the color of skin but also significant cosmetic concerns and
quality of life issues. Current treatment modalities, which include
topical corticosteroids, intralesional corticosteroids, phototherapy,
and systemic immunosuppression, are variably effective in
inducing repigmentation. Unfortunately, some cases of vitiligo
are refractory to treatment. There is a need for new, effective modalities
to treat patients with otherwise refractory vitiligo.
Needling is an office-based procedure that theoretically transposes
healthy, pigmented skin cells to depigmented areas using
a needle in vitiligo patients. Two preliminary studies of needling
as a novel treatment for vitiligo had promising results but were
limited by small sample size and subjective results.
The proposed RCT will further investigate the use of needling to
treat vitiligo. It differs from the previous studies in that it seeks
to identify the cause of clinical benefit by comparing needling
alone to needling with corticosteroid, examines a larger number
of patients, and quantifies improvement using CFM.

MELASMA
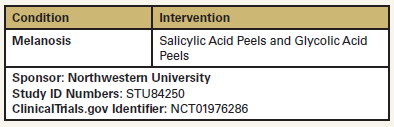
A Pilot Study Testing Salicylic Acid Peels Vs Glycolic Acid Peels for the Treatment of Melasma
The purpose of this study is to determine the safety and effectiveness
of glycolic acid chemical peels compared with salicylic
acid chemical peels for the treatment of melasma. Participants in
this study will be patients who are clinically diagnosed with at
least a 2 x 2 cm patch of melasma on each side of their face (forehead
or cheek). At baseline and at weeks 4, 8, and 12, one half
of the subject’s face will be randomly selected to receive 4 treatments
of 30% glycolic acid peels, and the other half of the face
will receive 4 treatments of 30% salicylic acid peels. The follow-up
visit will be at week 16. The change in best overall cosmetic
appearance (right side vs left side) will be rated by a blinded dermatologist
from baseline to week 16.