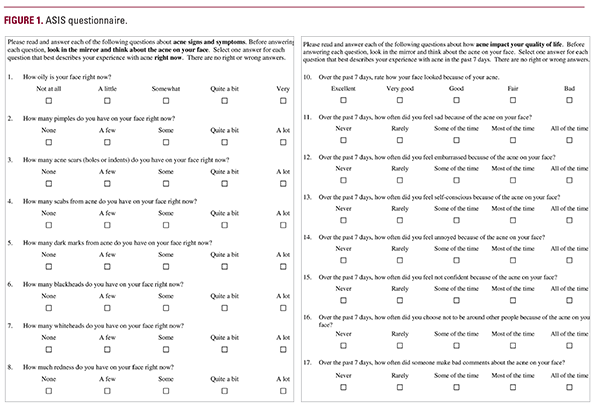
strated a positive impact of sarecycline. However, there have been no studies evaluating the effectiveness of sarecycline or any other oral antibiotics in AV through PROs in a real-world setting. PROs may be ideal for capturing the experience of patients with AV that is not adequately reflected by traditional clinical outcomes. Assessment of PROs is recommended in studies of patients with AV to complement standard outcomes reported and for use in routine clinical practice.24,25 Clinical trials in AV have employed a wide range of PROs, hampering the interpretation of research findings and comparisons across studies.26-28 PRO assessments in routine practice could complement clinical assessment and provide patients’ perspectives on treatment, including with oral antibiotics, as well as the impact of therapy on their physical, emotional, and psychosocial well-being. This prospective study evaluated PROs using a validated tool – the Acne Symptom and Impact Scale (ASIS) – and assessed the clinical effectiveness and safety of sarecycline in community practice settings across the US.
METHODS
Study Design and Participants
This single-arm, prospective, observational cohort study evaluated the use of sarecycline in patients with moderate-to-severe non-nodular AV (N=300) in the US. Data were collected from 30 community der-matology practices between March 2021 and May 2022. The study was approved by the Advarra Institutional Review Board (SSU00149823 and SSU00150552). Study patients were followed for up to 12 weeks with evaluations at weeks 4, 8, and 12.
This single-arm, prospective, observational cohort study evaluated the use of sarecycline in patients with moderate-to-severe non-nodular AV (N=300) in the US. Data were collected from 30 community der-matology practices between March 2021 and May 2022. The study was approved by the Advarra Institutional Review Board (SSU00149823 and SSU00150552). Study patients were followed for up to 12 weeks with evaluations at weeks 4, 8, and 12.
Patients ≥9 years old with a confirmed diagnosis of facial non-nodular moderate-to-severe AV (Investigator Global Assessment [IGA] score of 3 or 4) were included and received oral sarecycline (60 mg, 100 mg, or 150 mg). Appropriate sarecycline dosages were determined by the clini-cians based on clinical judgment per US FDA prescribing guidelines.21 Patients ≥18 years old provided informed written consent and those <18 years of age were consented to participate by their adult primary care-givers.
Patients were excluded if they had any facial conditions that interfered with AV clinical evaluations; had a history of allergy to tetracycline-class antibiotics or pseudomembranous or antibiotic-associated colitis; known resistance to other tetracyclines; were on concurrent treatment with penicillin or oral retinoids; were pregnant, lactating, or planning a pregnancy during the study period; or had inaccessible medical records.