To the Editor,
Glucagon-like peptide-1 receptor agonists (GLP1-RAs) are widely prescribed for type 2 diabetes and, more recently, for obesity management.2 With their expanding use, questions have emerged regarding perioperative safety. Delayed wound healing, fat necrosis, and tissue fragility have been described in breast reconstruction, and nausea and vomiting are welldocumented perioperative effects.1,2 To our knowledge, no large-scale study has evaluated postoperative outcomes in dermatologic surgery among patients using GLP1-RAs.
We conducted a retrospective cohort study using TriNetX, a federated health record network including over 130 million patients from more than 70 U.S. healthcare organizations.
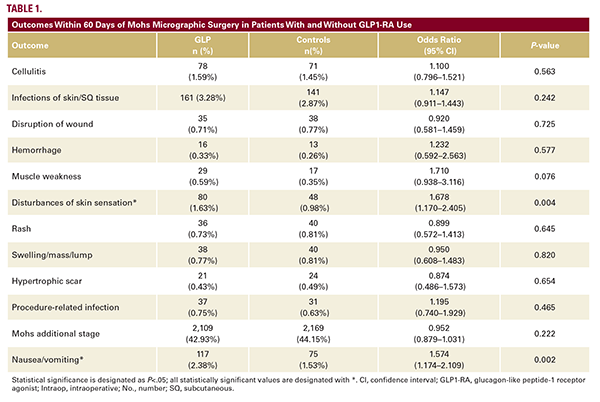
Adult patients undergoing Mohs micrographic surgery (MMS) between 2015 and 2025 were identified using CPT codes. Two cohorts were generated: patients with greater than or equal to 6 months of continuous GLP1-RA use prior to surgery, and patients with no history of GLP1-RA prescriptions. Six months of exposure was chosen based on existing literature suggesting that this threshold reflects peak GLP1-RA effects.3
One-to-one propensity score matching was used to balance cohorts by age, sex, race, BMI, diabetes status, cardiovascular risk factors, and immunosuppressive medication use. After matching, each cohort included 4,912 patients. Postoperative outcomes were identified within 60 days of MMS using ICD
Glucagon-like peptide-1 receptor agonists (GLP1-RAs) are widely prescribed for type 2 diabetes and, more recently, for obesity management.2 With their expanding use, questions have emerged regarding perioperative safety. Delayed wound healing, fat necrosis, and tissue fragility have been described in breast reconstruction, and nausea and vomiting are welldocumented perioperative effects.1,2 To our knowledge, no large-scale study has evaluated postoperative outcomes in dermatologic surgery among patients using GLP1-RAs.
We conducted a retrospective cohort study using TriNetX, a federated health record network including over 130 million patients from more than 70 U.S. healthcare organizations.
Adult patients undergoing Mohs micrographic surgery (MMS) between 2015 and 2025 were identified using CPT codes. Two cohorts were generated: patients with greater than or equal to 6 months of continuous GLP1-RA use prior to surgery, and patients with no history of GLP1-RA prescriptions. Six months of exposure was chosen based on existing literature suggesting that this threshold reflects peak GLP1-RA effects.3
One-to-one propensity score matching was used to balance cohorts by age, sex, race, BMI, diabetes status, cardiovascular risk factors, and immunosuppressive medication use. After matching, each cohort included 4,912 patients. Postoperative outcomes were identified within 60 days of MMS using ICD