INTRODUCTION
In 2021, the United States Food and Drug Administration (FDA) updated the boxed warnings for three JAK inhibitors (JAKi) following results from a post-marketing safety study of tofacitinib rheumatoid arthritis (RA) patients, which revealed increased risks of adverse events of special interest (AESIs) such as major adverse cardiovascular events (MACE) and malignancies.1,2 Baricitinib, one of the JAKis subject to the revised warning, was approved in 2022 for the treatment of alopecia areata (AA), a condition that typically affects a younger and healthier population. Given that the revised warnings were based on data from an RA cohort, there is a need for a comparative study discerning risks for AESIs between the two groups.3
MATERIALS AND METHODS
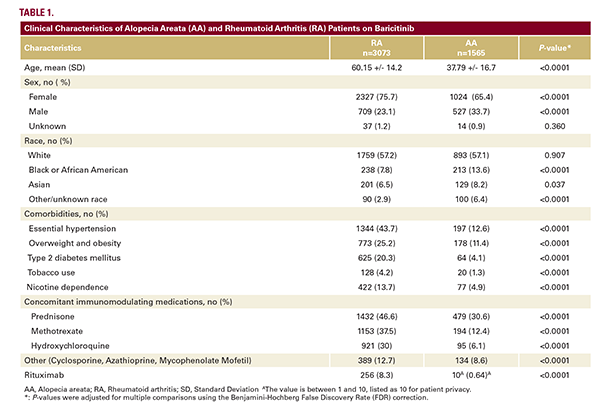
This retrospective cohort study utilized TriNetX, a global health research network of de-identified electronic health record data, to identify patients with a diagnosis of AA (ICD-10 L63) or RA (ICD-10 M05.9, M06) who were prescribed baricitinib between March 2006 and March 2025. Demographic and clinical characteristics were first evaluated in unmatched cohorts, then age-sex-race matching was performed to assess differences in AESI outcomes.
RESULTS
The RA cohort was older (mean age 60 vs 37.9 years), had a higher proportion of females (75.7% vs 65.4%), and exhibited a greater prevalence of comorbidities, including hypertension,