A large real world analysis identifies small but measurable perioperative signals for sensory change and nausea after Mohs micrographic surgery
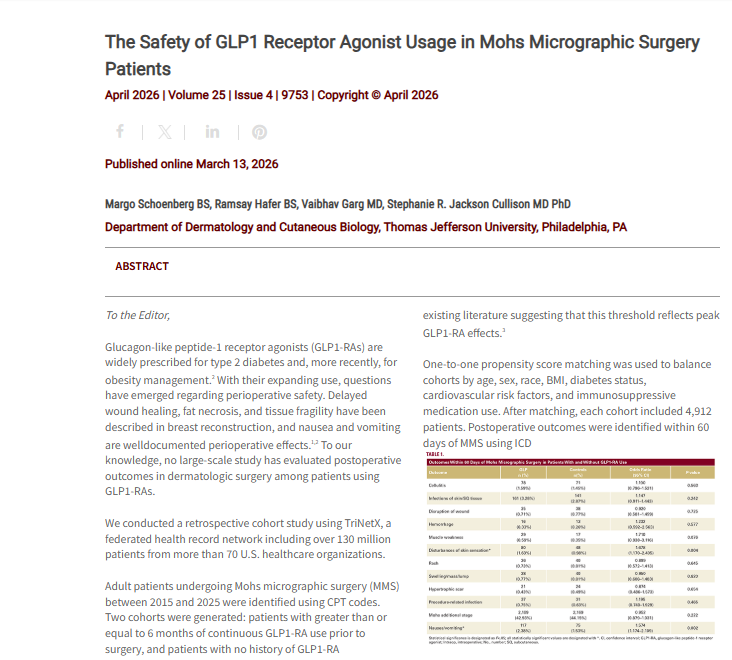
A retrospective cohort study using the TriNetX research network compared adults undergoing Mohs micrographic surgery who had at least six months of prior GLP-1 receptor agonist use with matched controls. After propensity score matching, each cohort included 4,912 patients balanced for age, sex, race, BMI, diabetes, cardiovascular risk factors, and immunosuppressive medication use.
The authors report that patients on GLP-1 receptor agonists were more likely to have postoperative disturbances of skin sensation and to experience postoperative nausea and vomiting within 60 days of surgery. No statistically significant differences were observed for wound infection, cellulitis, wound disruption, hemorrhage, hypertrophic scarring, need for additional Mohs stages, or several rare complications.
These findings describe associations in a large administrative dataset and do not establish causation. The increased nausea and vomiting aligns with known adverse effects of GLP-1 receptor agonists and supports the outcome detection approach. The sensory signal is novel and merits further investigation in prospective studies.
Read the full JDD article for cohort definitions, analytic methods, and subgroup data, and consider whether these observations should inform perioperative counseling and symptom management for patients receiving GLP-1 receptor agonists who are scheduled for dermatologic surgery.
J Drugs Dermatol. 2026;25(4). doi:10.36849/JDD.9753
Blog write-up assisted by AI