To the Editor,
Liquid Nitrogen (LN2) is one of the most common dermatologic procedures performed by dermatologists and other physicians. Actinic keratoses (AK), seborrheic keratoses (SK), and warts are the most common lesions treated with LN2. The 2023 CMS data and research statistics listed code 17110, destruction of benign lesions up to 14, and code 17000, destruction of premalignant lesions 1st, in the top 200 billed codes. Allowed services totaled 3,037,446 for code 17110 and 6,114,068 for code 17000 in 2023.1
To administer LN2, providers typically will hold a cryocanister in one hand while feeling for skin lesions with the other hand. Dermatologists often treat between 1 and 50 or more distinct sites per encounter, and scribes are often used to record the location, number, and diagnosis of each lesion. This current workflow inefficiency is associated with rapidly increasing labor costs and declining reimbursement. Without a scribe, providers rely on recall to document LN2 treatments. Visible cameras, as well as ambient artificial intelligence (AI) audio scribes, are of limited use in cryotherapy because they cannot accurately capture the locations of LN2-treated lesions (Figure 1). AI-scribes fail to address the practical needs of dermatology providers who spend a large portion of their visits administering LN2. We report a novel solution that enables providers to perform and document liquid nitrogen simultaneously.
Liquid Nitrogen (LN2) is one of the most common dermatologic procedures performed by dermatologists and other physicians. Actinic keratoses (AK), seborrheic keratoses (SK), and warts are the most common lesions treated with LN2. The 2023 CMS data and research statistics listed code 17110, destruction of benign lesions up to 14, and code 17000, destruction of premalignant lesions 1st, in the top 200 billed codes. Allowed services totaled 3,037,446 for code 17110 and 6,114,068 for code 17000 in 2023.1
To administer LN2, providers typically will hold a cryocanister in one hand while feeling for skin lesions with the other hand. Dermatologists often treat between 1 and 50 or more distinct sites per encounter, and scribes are often used to record the location, number, and diagnosis of each lesion. This current workflow inefficiency is associated with rapidly increasing labor costs and declining reimbursement. Without a scribe, providers rely on recall to document LN2 treatments. Visible cameras, as well as ambient artificial intelligence (AI) audio scribes, are of limited use in cryotherapy because they cannot accurately capture the locations of LN2-treated lesions (Figure 1). AI-scribes fail to address the practical needs of dermatology providers who spend a large portion of their visits administering LN2. We report a novel solution that enables providers to perform and document liquid nitrogen simultaneously.

The Technique
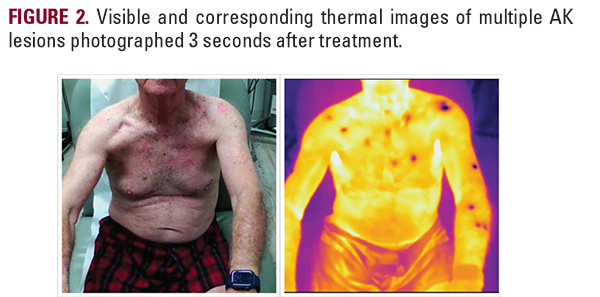
While visible imaging cannot capture LN2-treatment sites, thermal imaging can capture cryotherapy sites with precision (Figure 2). Cryotherapy-treated lesions appear as foci of cooler temperatures on thermal imaging and undergo temporal decay as they warm. When treating an AK for 10 seconds with a 1-2 mm rim of LN2, the AK will be visible on thermal imaging for approximately 1 minute after treatment.
We have developed a HIPAA-compliant solution that utilizes a lightweight adapter to attach an Android device with integrated visible and FLIR Lepton 3.5 thermal cameras to a cryocanister (Figure 3). After treating a region of lesions with LN2, the provider can quickly tap the camera screen, capturing visible and thermal images of the area. These images are processed via an advanced machine learning (ML) pipeline for automated treatment site detection and anatomical mapping. The provider can quick-select diagnoses, tag lesions to observe, and download a summary report that allows for electronic medical record (EMR) integration (Figure 4).
Practical Implications
Thermal imaging has been used to assess skin conditions, including psoriasis, skin cancers, burns, and cosmetic treatments such as cryolipolysis.2 To date, there are no reports on the use of thermal imaging for documentation of cryotherapy treatment sites. With current off-the-shelf thermal cameras, providers are unable to administer LN2 and subsequently operate a thermal camera quickly enough before treated lesions become undetectable. Incorporation of this novel thermal imaging
While visible imaging cannot capture LN2-treatment sites, thermal imaging can capture cryotherapy sites with precision (Figure 2). Cryotherapy-treated lesions appear as foci of cooler temperatures on thermal imaging and undergo temporal decay as they warm. When treating an AK for 10 seconds with a 1-2 mm rim of LN2, the AK will be visible on thermal imaging for approximately 1 minute after treatment.
We have developed a HIPAA-compliant solution that utilizes a lightweight adapter to attach an Android device with integrated visible and FLIR Lepton 3.5 thermal cameras to a cryocanister (Figure 3). After treating a region of lesions with LN2, the provider can quickly tap the camera screen, capturing visible and thermal images of the area. These images are processed via an advanced machine learning (ML) pipeline for automated treatment site detection and anatomical mapping. The provider can quick-select diagnoses, tag lesions to observe, and download a summary report that allows for electronic medical record (EMR) integration (Figure 4).
Practical Implications
Thermal imaging has been used to assess skin conditions, including psoriasis, skin cancers, burns, and cosmetic treatments such as cryolipolysis.2 To date, there are no reports on the use of thermal imaging for documentation of cryotherapy treatment sites. With current off-the-shelf thermal cameras, providers are unable to administer LN2 and subsequently operate a thermal camera quickly enough before treated lesions become undetectable. Incorporation of this novel thermal imaging








