To the Editor,
Rosacea is a chronic inflammatory disorder primarily affecting the face, characterized by erythema, telangiectasia, inflammatory lesions, and flushing.1 Rosacea affects up to 10% of the global population and is associated with a reduced quality of life and higher rates of depression and anxiety compared to the general population, underscoring the importance of effective treatment.1,2,3
Topicals, such as azelaic acid, metronidazole, sulfacetamide/ sulfur, brimonidine, ivermectin, and oxymetazoline, form the mainstay of rosacea treatment.1 Despite available therapies, access is often limited, and monthly out-of-pocket costs may exceed $100 in severe cases.4 Given the financial and psychosocial burdens of rosacea, this study examines formulary coverage of topical rosacea treatments among the ten largest US commercial health insurers.
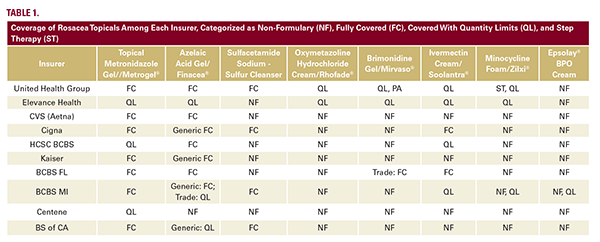
The top ten largest United States (US) commercial insurers by market share were identified from the American Medical Association’s 2024 Competition in Health Insurance report.5 Using 2025 formulary data, coverage of topical rosacea therapies was evaluated among these insurers and classified as full, with quantity limits (QL) or step therapy (ST) requirements, or nonformulary (NF) (Table 1).
As shown in Table 1, coverage varied widely across rosacea topicals. Metronidazole gel was the most consistently covered agent, included by all ten insurers, with three plans imposing QLs. Azelaic acid/Finacea® was fully covered by seven plans, and sulfacetamide sodium–sulfur cleanser by four.
In contrast, oxymetazoline hydrochloride cream (Rhofade®), brimonidine gel/ Mirvaso®, and ivermectin 1% cream (Soolantra®) were NF or QL restricted by most insurers. Newer therapies demonstrated minimal coverage, with minocycline foam (Zilxi®) only covered by one plan, and Epsolay® cream listed as NF across all plans.
Among the 10 largest US commercial insurers, QL or ST frequently restricted access to all rosacea topicals, and only one topical received universal coverage, highlighting substantial barriers to management.
Rosacea is a chronic inflammatory disorder primarily affecting the face, characterized by erythema, telangiectasia, inflammatory lesions, and flushing.1 Rosacea affects up to 10% of the global population and is associated with a reduced quality of life and higher rates of depression and anxiety compared to the general population, underscoring the importance of effective treatment.1,2,3
Topicals, such as azelaic acid, metronidazole, sulfacetamide/ sulfur, brimonidine, ivermectin, and oxymetazoline, form the mainstay of rosacea treatment.1 Despite available therapies, access is often limited, and monthly out-of-pocket costs may exceed $100 in severe cases.4 Given the financial and psychosocial burdens of rosacea, this study examines formulary coverage of topical rosacea treatments among the ten largest US commercial health insurers.
The top ten largest United States (US) commercial insurers by market share were identified from the American Medical Association’s 2024 Competition in Health Insurance report.5 Using 2025 formulary data, coverage of topical rosacea therapies was evaluated among these insurers and classified as full, with quantity limits (QL) or step therapy (ST) requirements, or nonformulary (NF) (Table 1).
As shown in Table 1, coverage varied widely across rosacea topicals. Metronidazole gel was the most consistently covered agent, included by all ten insurers, with three plans imposing QLs. Azelaic acid/Finacea® was fully covered by seven plans, and sulfacetamide sodium–sulfur cleanser by four.
In contrast, oxymetazoline hydrochloride cream (Rhofade®), brimonidine gel/ Mirvaso®, and ivermectin 1% cream (Soolantra®) were NF or QL restricted by most insurers. Newer therapies demonstrated minimal coverage, with minocycline foam (Zilxi®) only covered by one plan, and Epsolay® cream listed as NF across all plans.
Among the 10 largest US commercial insurers, QL or ST frequently restricted access to all rosacea topicals, and only one topical received universal coverage, highlighting substantial barriers to management.