INTRODUCTION
Severe acute respiratory syndrome corona virus2 (SARS-CoV-2) is the most recently identified member of the zoonotic pathogens of corona viruses. It started by causing an outbreak of pneumonia in December 2019 in Wuhan, China.1 Among all related acute respiratory syndromes (SARS-CoV, MERS CoV), SARS-CoV-2 remains to be the most infectious with the highest potential for human transmission and can eventually end up in acute respiratory distress syndrome (ARDS).2,3
Only 15% of COVID-19 cases progress to pneumonia and about 5% develop ARDS, septic shock, and/or multiple organ failure. The majority of cases only exhibit mild to moderate symptoms.4,5 A wide array of skin manifestations in COVID-19 infection were reported including maculopapular eruptions, morbilliform rashes, urticaria, chickenpox like lesions, livedo reticularis, covid toe, erythema multiforme, and pityriasis rosea, and several other patterns.6
Different speculations regarding cutaneous manifestations in COVID-19 emerged to explain their pathogenesis. Symptoms mimicking viral exanthems were thought to be an immune mediated response to the viral nucleotides while other cutaneous eruptions such as vasculitis and thrombophlebitis were recognized as a secondary systemic consequence to COVID-19 infection.7
Pityriasis rosea (PR) is a relatively common, self-limited papulo-squamous dermatosis of unknown origin, which mainly appears in adolescents and young adults (10–35 years) and is slightly more common in females.8 It has a sudden onset, and in its typical presentation, the eruption is preceded by a solitary patch termed “herald patchâ€, mainly located on the trunk. A few days later, secondary eruption appears, with small pink, oval macules, with a grayish peripheral scaling collarette around them. The secondary lesions adopt a characteristic distribution along the cleavage lines of the trunk, with a configuration of a “Christmas treeâ€.9
Only 15% of COVID-19 cases progress to pneumonia and about 5% develop ARDS, septic shock, and/or multiple organ failure. The majority of cases only exhibit mild to moderate symptoms.4,5 A wide array of skin manifestations in COVID-19 infection were reported including maculopapular eruptions, morbilliform rashes, urticaria, chickenpox like lesions, livedo reticularis, covid toe, erythema multiforme, and pityriasis rosea, and several other patterns.6
Different speculations regarding cutaneous manifestations in COVID-19 emerged to explain their pathogenesis. Symptoms mimicking viral exanthems were thought to be an immune mediated response to the viral nucleotides while other cutaneous eruptions such as vasculitis and thrombophlebitis were recognized as a secondary systemic consequence to COVID-19 infection.7
Pityriasis rosea (PR) is a relatively common, self-limited papulo-squamous dermatosis of unknown origin, which mainly appears in adolescents and young adults (10–35 years) and is slightly more common in females.8 It has a sudden onset, and in its typical presentation, the eruption is preceded by a solitary patch termed “herald patchâ€, mainly located on the trunk. A few days later, secondary eruption appears, with small pink, oval macules, with a grayish peripheral scaling collarette around them. The secondary lesions adopt a characteristic distribution along the cleavage lines of the trunk, with a configuration of a “Christmas treeâ€.9
CASE
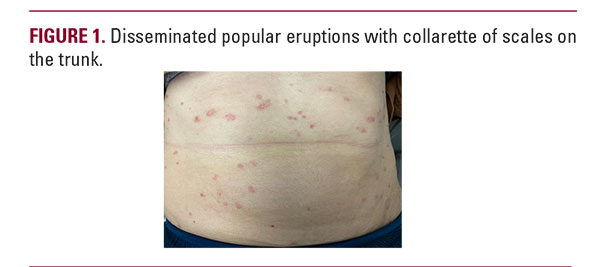
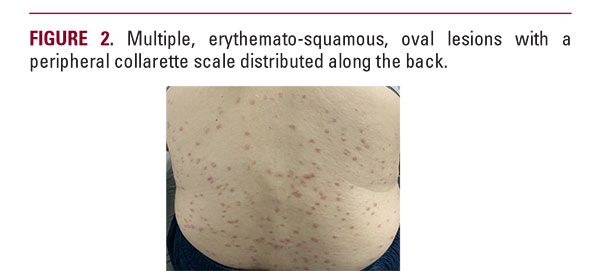
A 33-year-old female patient presented to the dermatology clinic with fever and a pruritic skin rash. The patient was fatigued and gave a history of cervical lymphadenopathy and shortness of breath. Upon skin examination, a rash consisting of oval-shaped salmon-colored patches and papules, up to 2cm in diameter, surrounded by light white scales located on her, chest, back thighs and upper limbs was noticed (Figures 1–2) She described that the lesions erupted 10 days before showing up to clinic. It started by an erythematous and scaly annular plaque that appeared on the left forearm and was accompanied by a low-grade fever and bouts of gastroenteritis that she was not concerned about. Several days later, the lesions continued to disseminate and became pruritic until she attended the clinic. Her oral and ocular mucosae were free of any presentations. The patient used a “mometasone furoate 0.1%” cream, which was prescribed to her by a local pharmacist with no response before attending clinic. No relevant medical history was given by the patient. Laboratory investigations showed lymphopenia. Chest x-ray and a positive nasopharyngeal smear test were consistent with COVID-19 infection. The patient confirmed that to her knowledge she had not been exposed to anyone who was diagnosed with COVID-19, however she works as a local seller in a traditional market with exposure to hundreds of people every day.