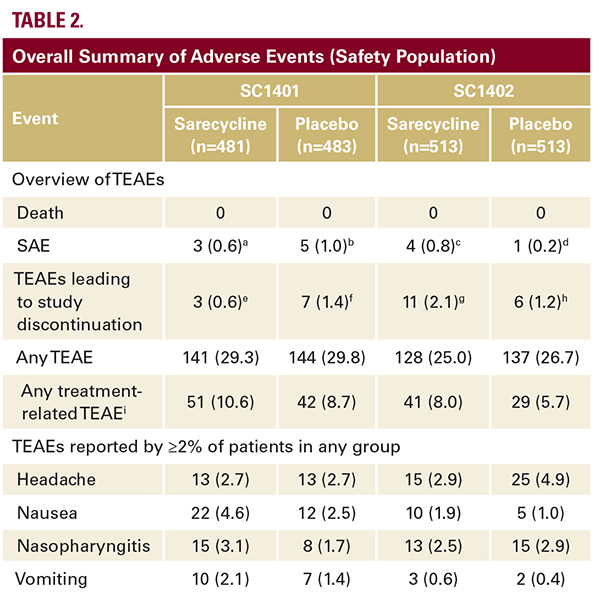
 TEAE severity was mild or moderate in most patients in both studies; 95.9% (233/243) of TEAEs in the sarecycline group and 96.0% (238/248) in the placebo group in study SC1401 were mild or moderate, as were 97.8% (224/229) of TEAEs in the sarecycline group and 97.0% (226/233) in the placebo group in study SC1402.Serious adverse events were rare in both studies, and all were considered not related or possibly related to study treatment. There were no deaths during either study. There were no clinically meaningful differences between the sarecycline and placebo groups in clinical laboratory, vital sign, and ECG measurements in either study.Adverse Events Leading to Discontinuations In study SC1401, TEAEs leading to study discontinuations occurred in 0.6% (3/481) and 1.4% (7/483) of patients in the sarecycline and placebo groups, respectively, but none were judged by the investigator as being related to study treatment; most were judged as possibly related (Table 2). In study SC1402, 2.1% (11/513) and 1.2% (6/513) of patients in the sarecycline and placebo groups, respectively, discontinued due to AEs, the majority of which were judged by the investigator as possibly related or related to study treatment.
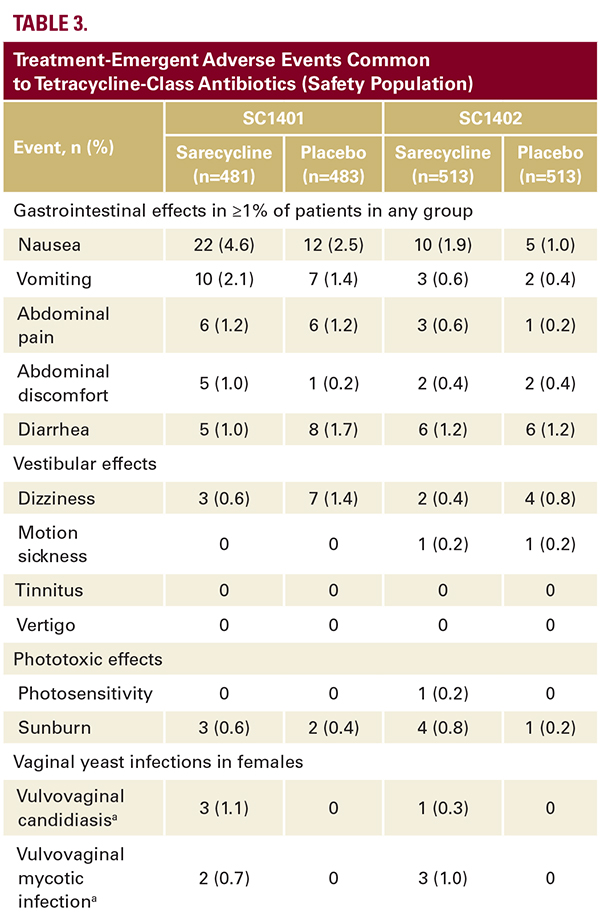
TEAE severity was mild or moderate in most patients in both studies; 95.9% (233/243) of TEAEs in the sarecycline group and 96.0% (238/248) in the placebo group in study SC1401 were mild or moderate, as were 97.8% (224/229) of TEAEs in the sarecycline group and 97.0% (226/233) in the placebo group in study SC1402.Serious adverse events were rare in both studies, and all were considered not related or possibly related to study treatment. There were no deaths during either study. There were no clinically meaningful differences between the sarecycline and placebo groups in clinical laboratory, vital sign, and ECG measurements in either study.Adverse Events Leading to Discontinuations In study SC1401, TEAEs leading to study discontinuations occurred in 0.6% (3/481) and 1.4% (7/483) of patients in the sarecycline and placebo groups, respectively, but none were judged by the investigator as being related to study treatment; most were judged as possibly related (Table 2). In study SC1402, 2.1% (11/513) and 1.2% (6/513) of patients in the sarecycline and placebo groups, respectively, discontinued due to AEs, the majority of which were judged by the investigator as possibly related or related to study treatment. Adverse Events Reported with Other Tetracycline-Class Antibiotics Among AEs reported with other tetracycline-class antibiotics, vestibular TEAEs (specifically dizziness, vertigo, tinnitus) and phototoxic TEAEs (photosensitivity, sunburn) were rare in sarecycline-treated patients, occurring in ≤ 1% of patients, and rates of GI TEAEs for sarecycline were low (Table 3). In each study, there were no cases of vertigo or tinnitus, and fewer cases of dizziness in sarecycline-treated patients than in placebo-treated patients. The most common GI TEAEs were nausea, vomiting, diarrhea, abdominal pain, abdominal discomfort, and constipation.
Adverse Events Reported with Other Tetracycline-Class Antibiotics Among AEs reported with other tetracycline-class antibiotics, vestibular TEAEs (specifically dizziness, vertigo, tinnitus) and phototoxic TEAEs (photosensitivity, sunburn) were rare in sarecycline-treated patients, occurring in ≤ 1% of patients, and rates of GI TEAEs for sarecycline were low (Table 3). In each study, there were no cases of vertigo or tinnitus, and fewer cases of dizziness in sarecycline-treated patients than in placebo-treated patients. The most common GI TEAEs were nausea, vomiting, diarrhea, abdominal pain, abdominal discomfort, and constipation.DISCUSSION
Sarecycline is the first narrow-spectrum tetracycline-class antibiotic for the treatment of moderate to severe acne. These pivotal phase 3 studies demonstrated that oral sarecycline 1.5 mg/kg per day for 12 weeks was effective in the treatment of moderate to severe acne vulgaris, with an onset of efficacy for inflammatory lesions observed as early as the first follow-up visit at week 3, and an overall safety profile generally similar to that of placebo. In addition, sarecycline showed a significant effect on acne severity in nonfacial sites.Interestingly, sarecycline showed a statistically significantly greater improvement than placebo in noninflammatory lesion counts beginning at week 6 in study SC1401 and week 9 in study SC1402, with continued improvement through week 12. The analyses in patients with at least 10 noninflammatory lesions at baseline provided further evidence of the therapeutic effect on noninflammatory lesions. Although the exact effect of sarecycline on noninflammatory lesions is unknown, a possible






