be applied to gene expression data in order to predict cSCC
outcomes and predict high-risk patients. A cSCC gene expression
profile test (cSCC-GEP) has been validated on residual
tumor from primary cSCC biopsies and can classify lesions as
either Class 1 (low risk) or Class 2 (high risk). The goal of this
study was to determine the impact of this genomic technology
on the clinical management of cSCC patients.
METHODS
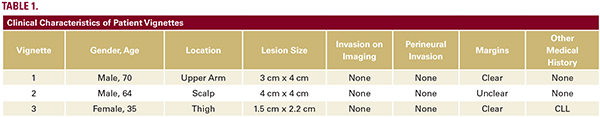
Dermatologists who attended a national Dermatology conference completed an online survey with three cSCC patient vignettes. Each vignette presented an image and described patient characteristics such as age, gender, and anatomic location of lesion, as well as lesion characteristics (e.g. tumor size, margin of resection, invasion of bone, and perineural invasion). (Table 1) Respondents answered a series of questions using pre and post-test methodology to evaluate the effect of Class 1 (low risk) and Class 2 (high risk) cSCC-GEP test results on management (adjuvant radiation therapy, chemotherapy/immunotherapy, referral for SLNBx, and recommended length of follow-up interval [q3 months, q6 months, and q12 months]).
The primary outcomes were the proportion of respondents who would alter their decision to perform a SLNBx or recommend adjuvant radiation therapy and/or chemotherapy/immunotherapy with the addition of cSCC-GEP test results, as well as the proportion who would alter their recommended follow-up interval. McNemar’s test was performed for assessing categorical variables, and the Wilcoxon signed-rank test was performed for assessing ordinal variables. Clinical recommendations for Class 1 and Class 2 cSCC-GEP test results were compared to baseline (without cSCC-GEP test result).
RESULTS
Respondent and Patient Vignette Characteristics
Overall, 435 dermatologists participated in the survey with a completion rate of 92.4% (N=402). Of the respondents, 33% reported being in practice for 11-20 years followed by 28% for 1-10 years and 20% for 21-30 years. (Table 2) Over 50% of dermatologists reported seeing an average of 1-5 high-risk cSCC patients per year and almost 25% of dermatologists reported seeing greater than 10 high-risk cSCC patients per year. The majority of respondents (>60%) reported using the AJCC system to stage cSCC, however over 35% of dermatologists reported not using a staging system at all.
Impact of cSCC-GEP Test Results on Decision to Recommend Adjuvant Radiation Therapy
A Class 1 cSCC-GEP test result led to a statistically significant decrease in recommended adjuvant radiation therapy by dermatologists in all vignettes. (Table 3) For all vignettes with a Class 2 cSCC-GEP test result, there was a statistically significant increase in adjuvant radiation therapy recommendations.
Impact of cSCC-GEP Test Results on Decision to Recommend Adjuvant Chemotherapy or Immunotherapy
A Class 1 cSCC-GEP test result led to a statistically significant decrease in recommended adjuvant chemotherapy or immu-

Overall, 435 dermatologists participated in the survey with a completion rate of 92.4% (N=402). Of the respondents, 33% reported being in practice for 11-20 years followed by 28% for 1-10 years and 20% for 21-30 years. (Table 2) Over 50% of dermatologists reported seeing an average of 1-5 high-risk cSCC patients per year and almost 25% of dermatologists reported seeing greater than 10 high-risk cSCC patients per year. The majority of respondents (>60%) reported using the AJCC system to stage cSCC, however over 35% of dermatologists reported not using a staging system at all.
Impact of cSCC-GEP Test Results on Decision to Recommend Adjuvant Radiation Therapy
A Class 1 cSCC-GEP test result led to a statistically significant decrease in recommended adjuvant radiation therapy by dermatologists in all vignettes. (Table 3) For all vignettes with a Class 2 cSCC-GEP test result, there was a statistically significant increase in adjuvant radiation therapy recommendations.
Impact of cSCC-GEP Test Results on Decision to Recommend Adjuvant Chemotherapy or Immunotherapy
A Class 1 cSCC-GEP test result led to a statistically significant decrease in recommended adjuvant chemotherapy or immu-







